Reading Reactive Surfaces in Motion
Open field notes from the Reactive Patinas™ studio table.
D. Mune
1/27/20262 min read
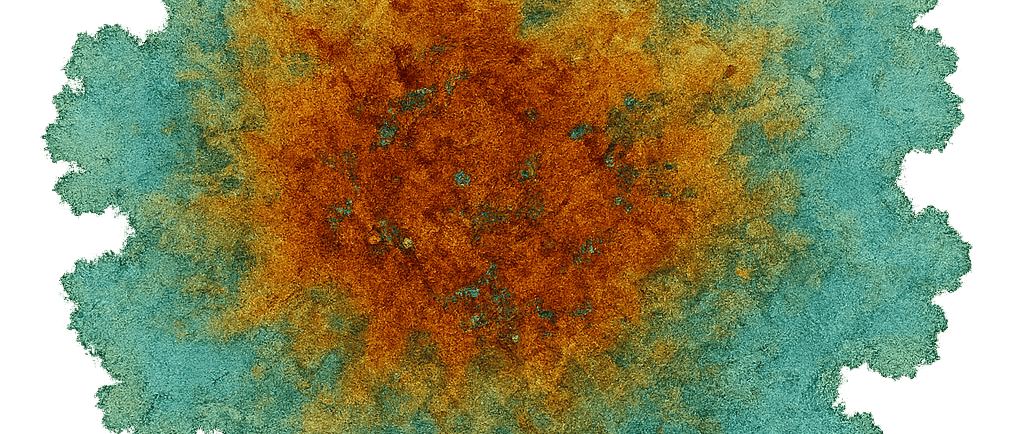
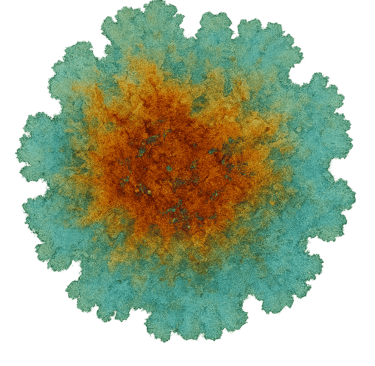
Table Talk is a slow, technical conversation about reactive materials as they actually behave —not as they are supposed to behave
These posts are not tutorials.
They are observations, interpretations, and field logic pulled directly from active experiments.
Each Table Talk focuses on one surface, one moment, or one anomaly — and asks a single question:
What is the material telling us, if we listen carefully enough?
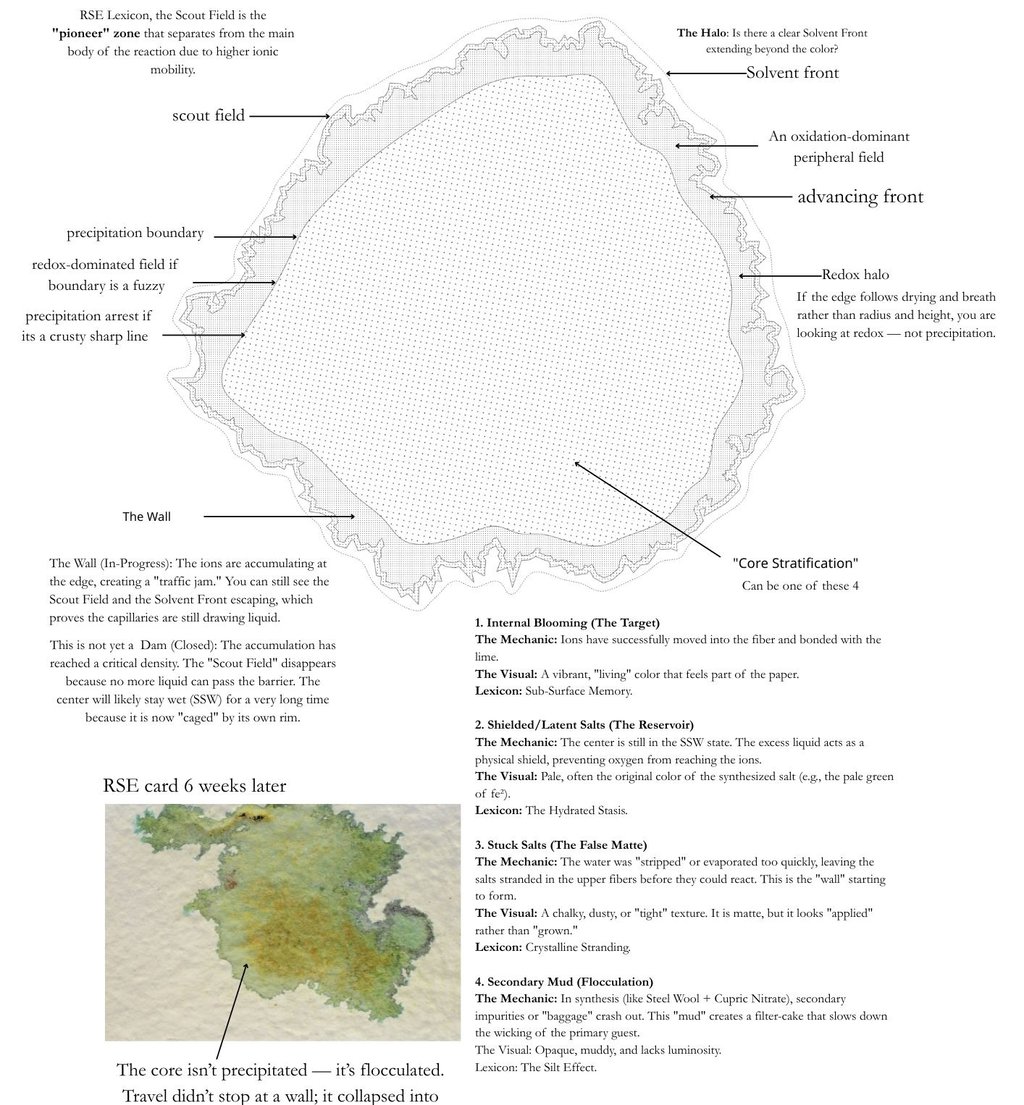
What You're Looking At
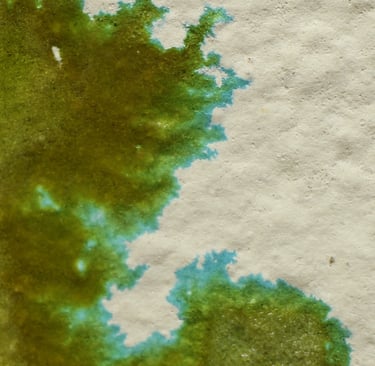
“An RSE Card . Reactive paper onto which Nickel Nitrate solution at 50% saturation was pooled”
Nickel Nitrate RSE — Reading the Blue-Green Halo
What you’re looking at A nickel nitrate RSE card showing a pale blue-green halo advancing ahead of the main olive core. The halo is detached from the bulk stain and rides the outer capillary edge.
What’s actually happening This is a Law of Travel effect, not a surface anomaly.
The water front outruns the nickel ion mass. Nitrate (NO₃⁻), with low hydration drag and high mobility, rides the capillary wave outward first. Nickel (Ni²⁺), burdened by its hydration shell and early surface interactions, lags behind.
The result is a temporary nitrate-rich / nickel-poor frontier — a chemical environment that can only exist in motion.
At that frontier:
pH shifts before nickel arrives in force
oxygen availability is highest
the moisture film is thinnest
When nickel finally enters this zone, it coordinates differently than it does in the core pool.
What's Actually Happening
What’s Actually Happening
The water front outruns the nickel ion mass. Nitrate (NO₃⁻), with low hydration drag and high mobility, rides the capillary wave outward first. Nickel (Ni²⁺), burdened by its hydration shell and early surface interactions, lags behind.
The result is a temporary nitrate-rich / nickel-poor frontier — a chemical environment that can only exist in motion.
At that frontier:
pH shifts before nickel arrives in force
oxygen availability is highest
the moisture film is thinnest
When nickel finally enters this zone, it coordinates differently than it does in the core pool.
Key rule:
This is not a surface defect. It is a Law of Travel event recorded in place.
Why The Surface Looks This Way
Why the halo is blue-green Nickel does not rust like iron, but it does shift coordination state depending on:
hydration
hydroxide availability
oxygen exposure
At the advancing edge, nickel forms lower-density, hydrated hydroxide / oxide complexes that favor blue-green tones. In the center, where moisture is thicker and oxygen is limited, the system settles into olive and sage states
This is redox and coordination timing, not precipitation.
Why This Shows Up Clearly Here
Why this shows up so clearly in RSE. RSE strips away bulk mixing:
Paper enforces radial flow
There is no convection to remix ions
Evaporation arrests the reaction mid-motion
In cement or plaster, this separation often collapses back into the pool .In RSE, the surface records the moment of separation.
Key distinction:
In bulk systems, this collapses.In reactive surfaces, it records.
What Would Change This Outcome
What would change this behavior
Higher humidity would soften or widen the halo
Pre-alkalinity would push the frontier outward
Oxidizers could collapse the separation or sharpen it
Cobalt or copper nitrates show related but distinct edge behavior due to different hydration and redox kinetics
Field Notes
Travel did not stop — it reorganized.
The edge is not decoration; it is a boundary condition.
You can overwhelm entry. You cannot outrun time.
#LawOfEntry#LawOfTravel#IonicFrontSeparation#CapillaryLag#NickelCoordination#RSEObserved






2106 House Ave, Cheyenne, Wyoming ·